Naveed A
Level 2
ISO, Regulatory Affairs FDA, EU, UK, PMN 510k, QMS and Professionalism
Habilidades

Conheça meus serviços

Quer trabalhar com remuneração por hora?
Diga a Naveed A o que você precisa.
US$ 100
/
horaPortfólio
Experiência profissional
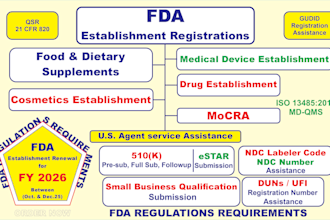
Senior Regulatory Affairs & QMS Consultant | EU MDR, FDA 510(k), ISO 13485
MDR Advisory • Freelance
Oct 2022 - Present • 3 yrs 7 mos
Regulatory Affairs & QMS Consultant with 25+ years of experience supporting clients across medical devices, IVDs, cosmetics, food, and pharmaceuticals. Specialized in EU MDR, UKCA, and FDA compliance, including 510(k) submissions and FDA Establishment Registration. Key achievements include preparation of Technical Documentation/Technical Files, development of ISO 13485 QMS documentation, and execution of risk management (ISO 14971), CER, and IFU compliance. Conducted gap assessments, internal audits, and provided practical regulatory strategies to ensure audit readiness and efficient market access. Focused on delivering clear, compliant, and actionable solutions tailored to client needs.
91 Avaliações
| (86) | ||
| (2) | ||
| (1) | ||
| (0) | ||
| (2) |
Classificação detalhada
- Nível de comunicação do freelancer
- Qualidade da entrega
- Valor da entrega
Ordenar por
dolivierp
Cliente recorrente

Argentina

ilanohayon443
Cliente recorrente

Estados Unidos

lukehunter318
Cliente recorrente

Estados Unidos
My 3rd year in a row working with him, always a pleasure.
Resposta do freelancer
jasonwrightevo
Cliente recorrente

Reino Unido
Resposta do freelancer
jasonwrightevo
Cliente recorrente

Reino Unido